Sinopharm’s Vaccine Nears Emergency-Use Approval by WHO
By Wang Lijun and Denise Jia
May 05, 2021 03:49 AM
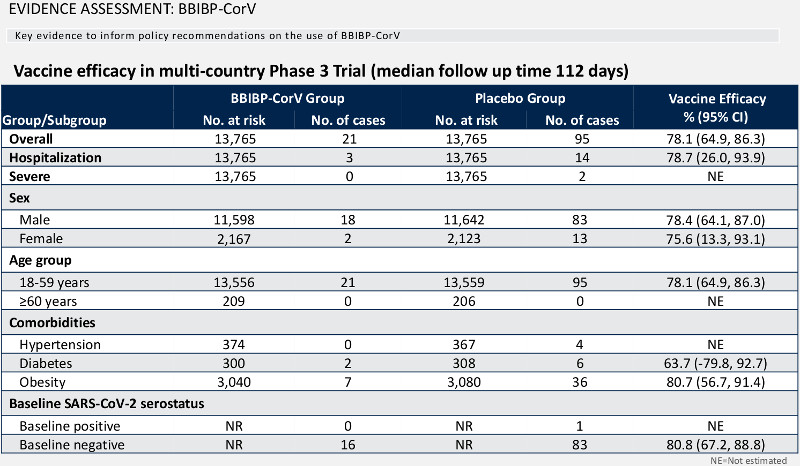
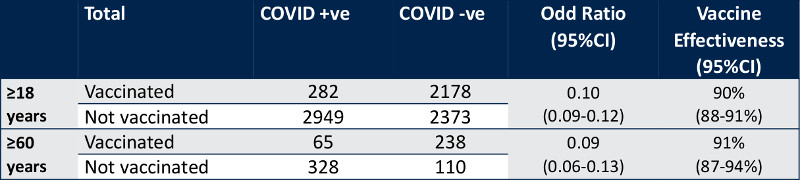
World Health Organization experts said one of China' Covid-19 vaccines is 78.1% effective in adults ages 18-59 years old but found that there is insufficient trial data to demonstrate effectiveness in protecting older people and people with underlying medical conditions.
The United Nations organization's Strategic Advisory Group of Expert on Immunization published the assessment in a report on BBIBP-CorV, the Covid-19 vaccine developed by state owned China National Pharmaceutical Group Corp. (Sinopharm). The finding could play a key role in approval by the World Health Organization (WHO) of Sinopharm' vaccine for emergency use.
..........
Related link: Sinopharm’s Vaccine Nears Emergency-Use Approval by WHO